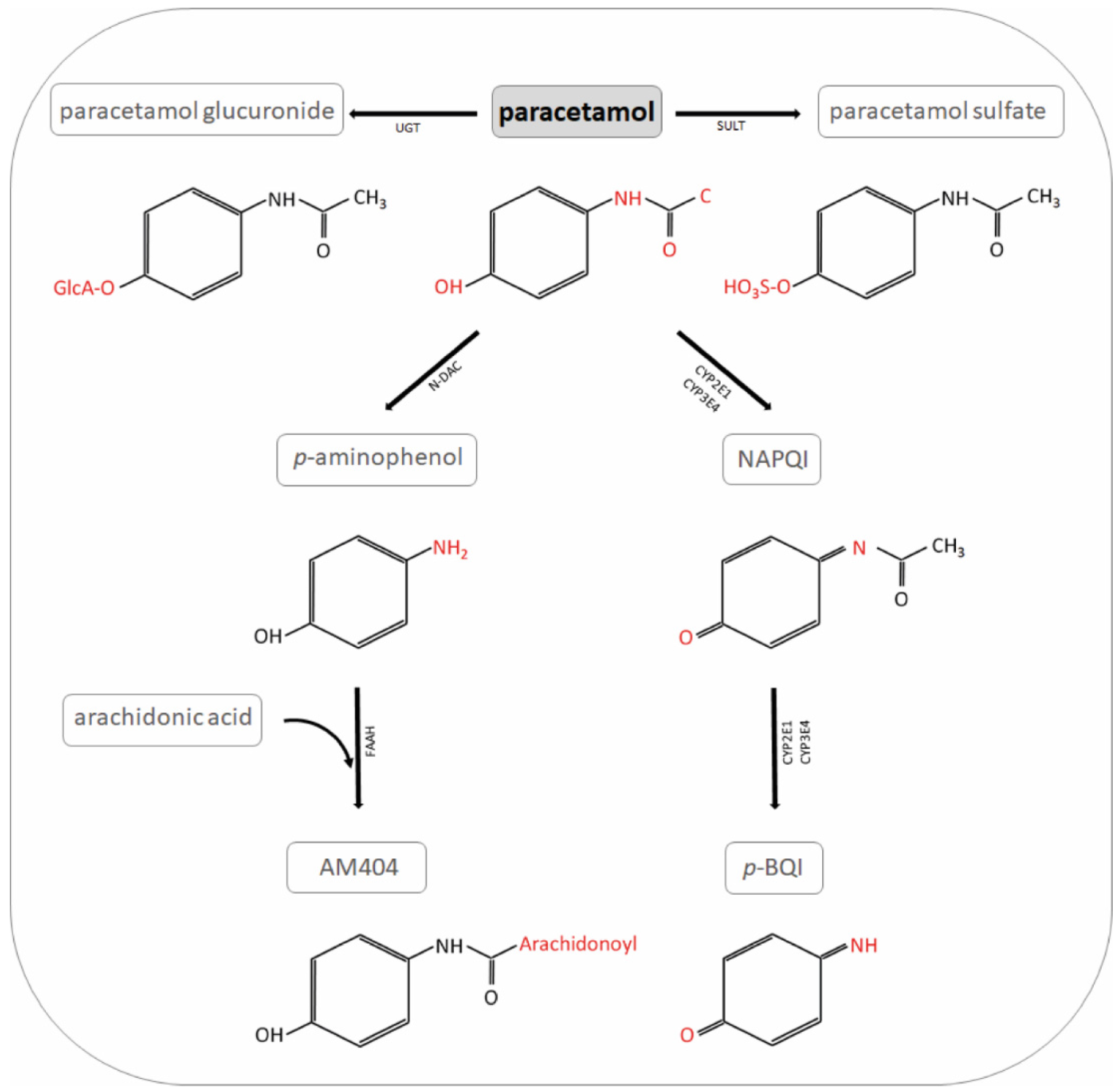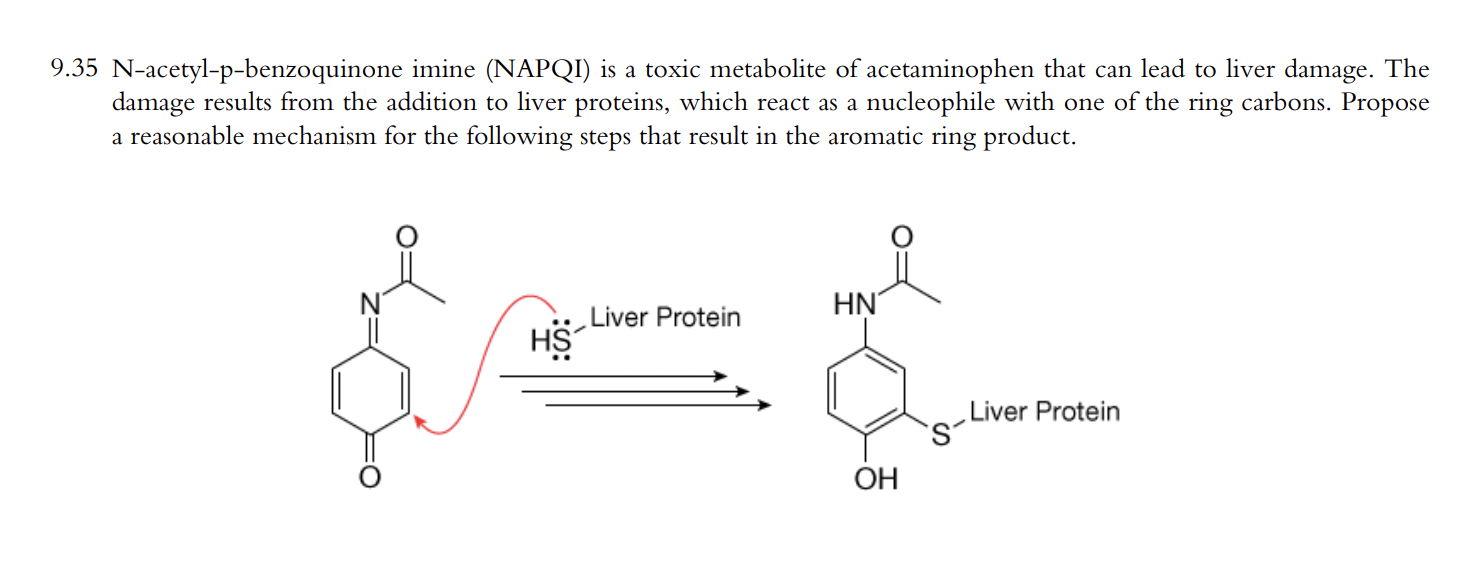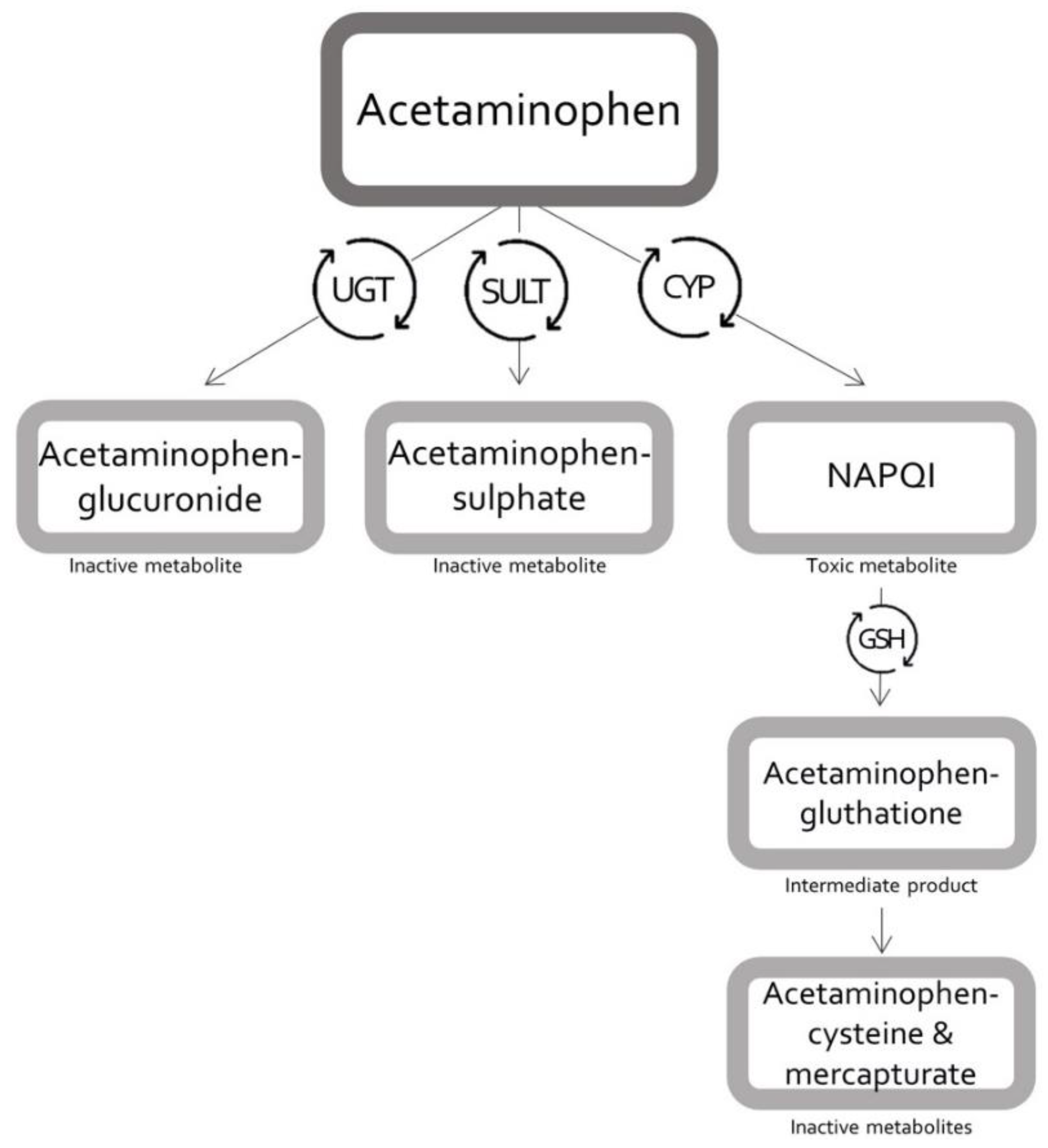
Pharmaceutics | Free Full-Text | Modelling Tools to Characterize Acetaminophen Pharmacokinetics in the Pregnant Population
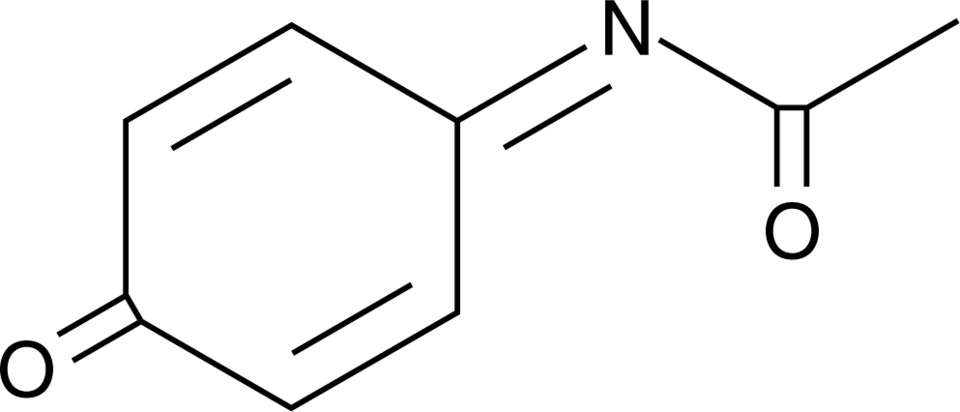
N-Acetyl-4-benzoquinone imine (NAPQI, N-Acetyl-p-benzoquinonimine, CAS Number: 50700-49-7) | Cayman Chemical

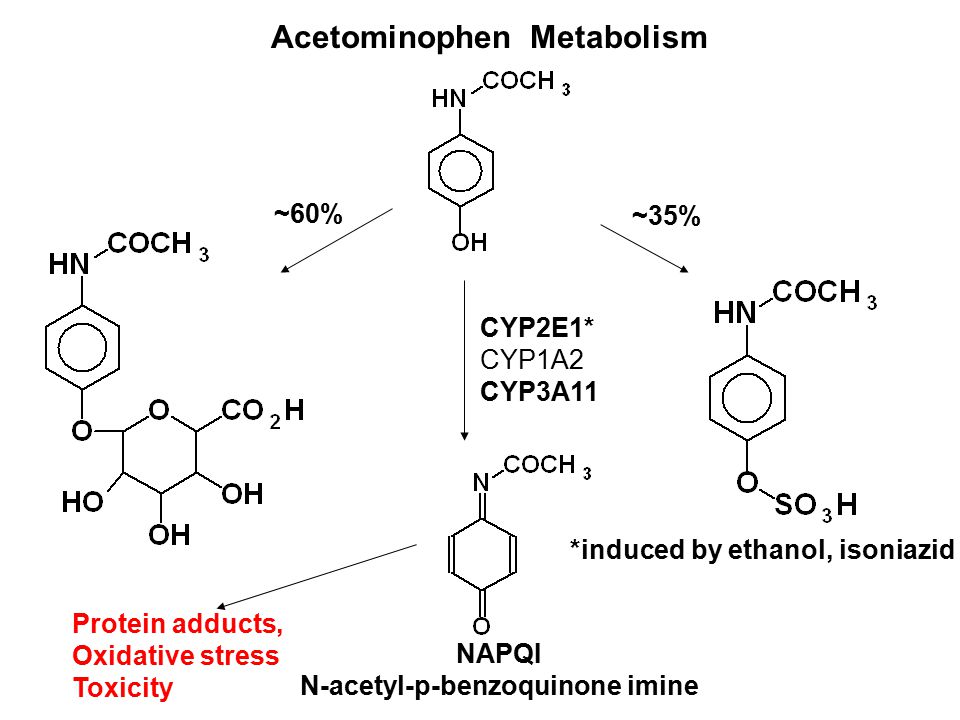
Acetaminophen metabolism. NAPQI, N-acetyl-p-benzoquinone imine; CYP450,... | Download Scientific Diagram

Tunable oxidative release of N-acetyl-p-benzoquinone-imine and acetamide from electrochemically derived sub-monolayer acetaminophen modified glassy carbon electrode - ScienceDirect

PDF) N-Acetyl-p-benzoquinone imine (NAPQI) induces Ca2+ release from mitochondria by stimulating pyridine nucleotide hydrolysis

LC-MS analyses of N-acetyl-p-benzoquinone imine-adducts of glutathione, cysteine, N-acetylcysteine, and albumin in a plasma sample: A case study from a patient with a rare acetaminophen-induced acute swelling rash. | Semantic Scholar

What Are the Potential Sites of Protein Arylation by N-Acetyl-p-benzoquinone Imine (NAPQI)? | Chemical Research in Toxicology

Transformation of Acetaminophen by Chlorination Produces the Toxicants 1 , 4 -Benzoquinone and N-Acetyl-p-benzoquinone Imine | Semantic Scholar

N-Acetyl-p-benzoquinone Imine, the Toxic Metabolite of Acetaminophen, Is a Topoisomerase II Poison | Biochemistry

In-source formation of N-acetyl-p-benzoquinone imine (NAPQI), the putatively toxic acetaminophen (paracetamol) metabolite, after derivatization with pentafluorobenzyl bromide and GC–ECNICI-MS analysis - ScienceDirect

Transformation of Acetaminophen by Chlorination Produces the Toxicants 1 , 4 -Benzoquinone and N-Acetyl-p-benzoquinone Imine | Semantic Scholar