Treatment Algorithms for Crohn's Disease - FullText - Digestion 2020, Vol. 101, Suppl. 1 - Karger Publishers
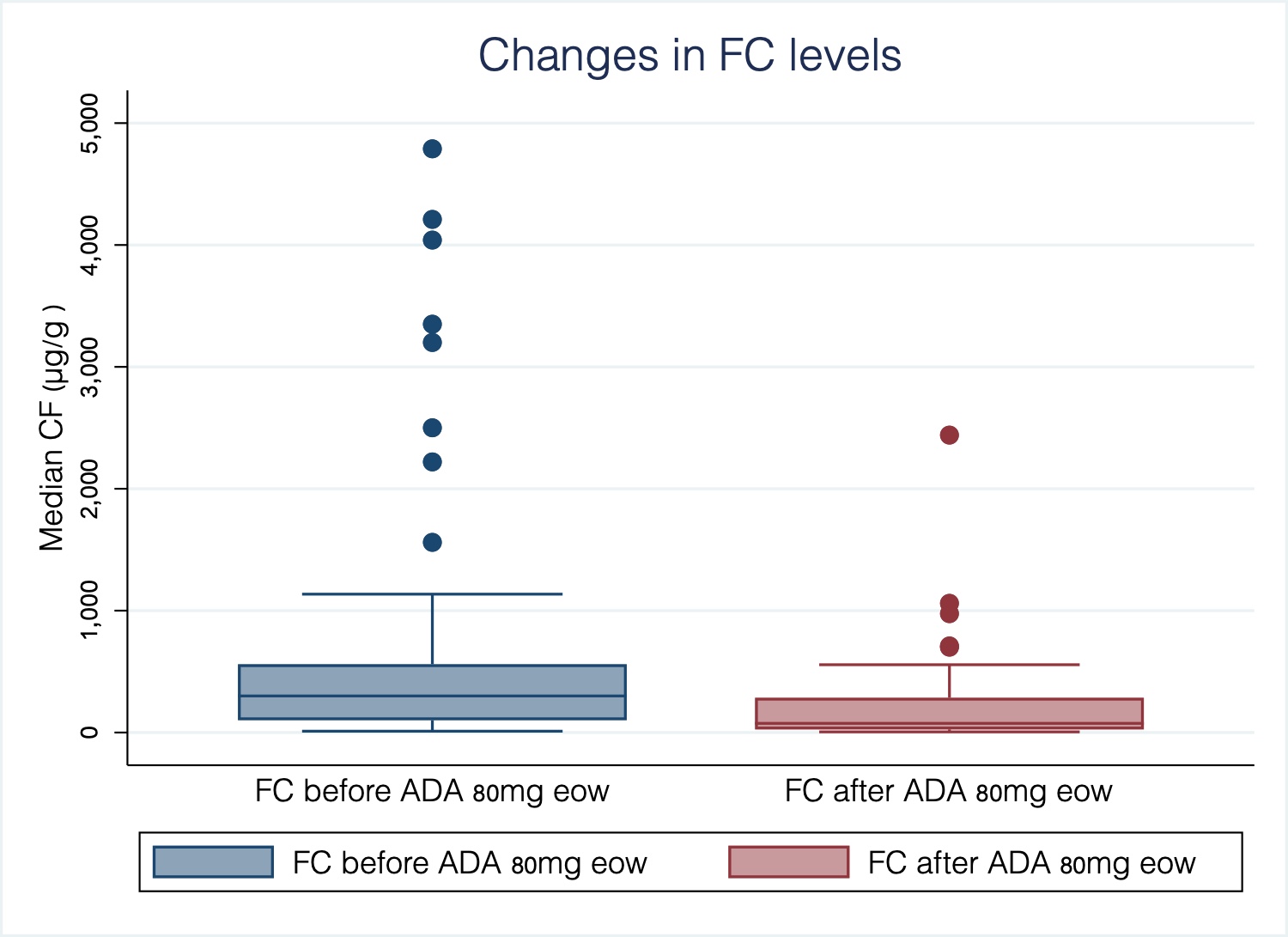
European Crohn´s and Colitis Organisation - ECCO - P533 Adalimumab 80mg every other week in inflammatory bowel disease: Treatment intensification outcomes in real life clinical practice
![PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/434bc5078bbc62a1a4782f27592fcf28f239679e/9-Table3-1.png)
PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar
![PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/434bc5078bbc62a1a4782f27592fcf28f239679e/8-Table1-1.png)
PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar
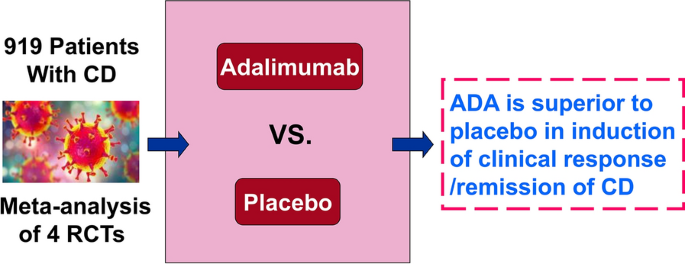
Adalimumab for induction of remission in patients with Crohn's disease: a systematic review and meta-analysis | European Journal of Medical Research | Full Text

Adalimumab biosimilars, ABP501 and SB5, are equally effective and safe as adalimumab originator | Scientific Reports
![PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/434bc5078bbc62a1a4782f27592fcf28f239679e/8-Table2-1.png)
PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar

PDF) P553 Efficacy, safety and cost-efficiency of adalimumab 80 mg every other week in previously intensified IBD patients under treatment with adalimumab 40 mg every week
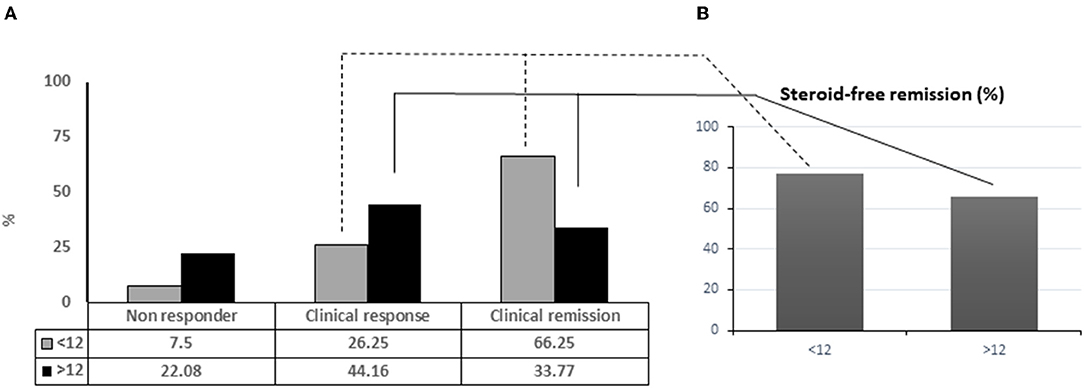
Frontiers | Administration Timing Is the Best Clinical Outcome Predictor for Adalimumab Administration in Crohn's Disease

Increased versus conventional adalimumab dose interval for patients with Crohn's disease in stable remission (LADI): a pragmatic, open-label, non-inferiority, randomised controlled trial - The Lancet Gastroenterology & Hepatology

PDF) Efficacy and Safety of Adalimumab by Disease Duration: Analysis of Pooled Data From Crohn's Disease Studies

The Toronto Consensus Statements for the Management of Inflammatory Bowel Disease in Pregnancy - Gastroenterology

PDF) Adalimumab Monotherapy and a Combination with Azathioprine for Crohn's Disease: A Prospective, Randomized Trial

European Crohn´s and Colitis Organisation - ECCO - P533 Adalimumab 80mg every other week in inflammatory bowel disease: Treatment intensification outcomes in real life clinical practice